 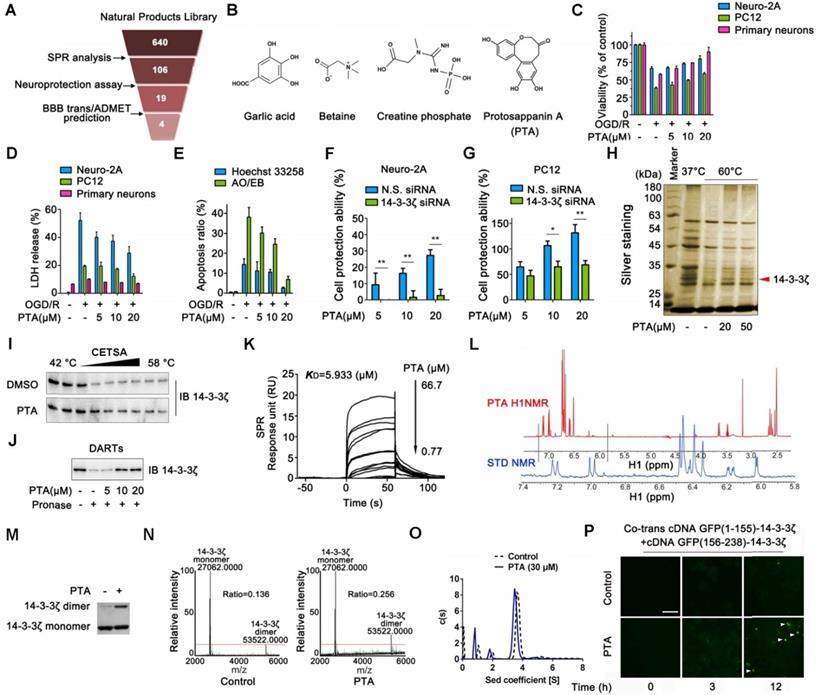
The first era of targeted protein degradation (TPD) began with publication of the pivotal proteolysis-targeting chimera (PROTAC) paper by Sakamoto et al. CMR, calculated molecular refractivity tPSA, total polar surface area. The pros and cons of these modalities are discussed in the text and in Box 1. 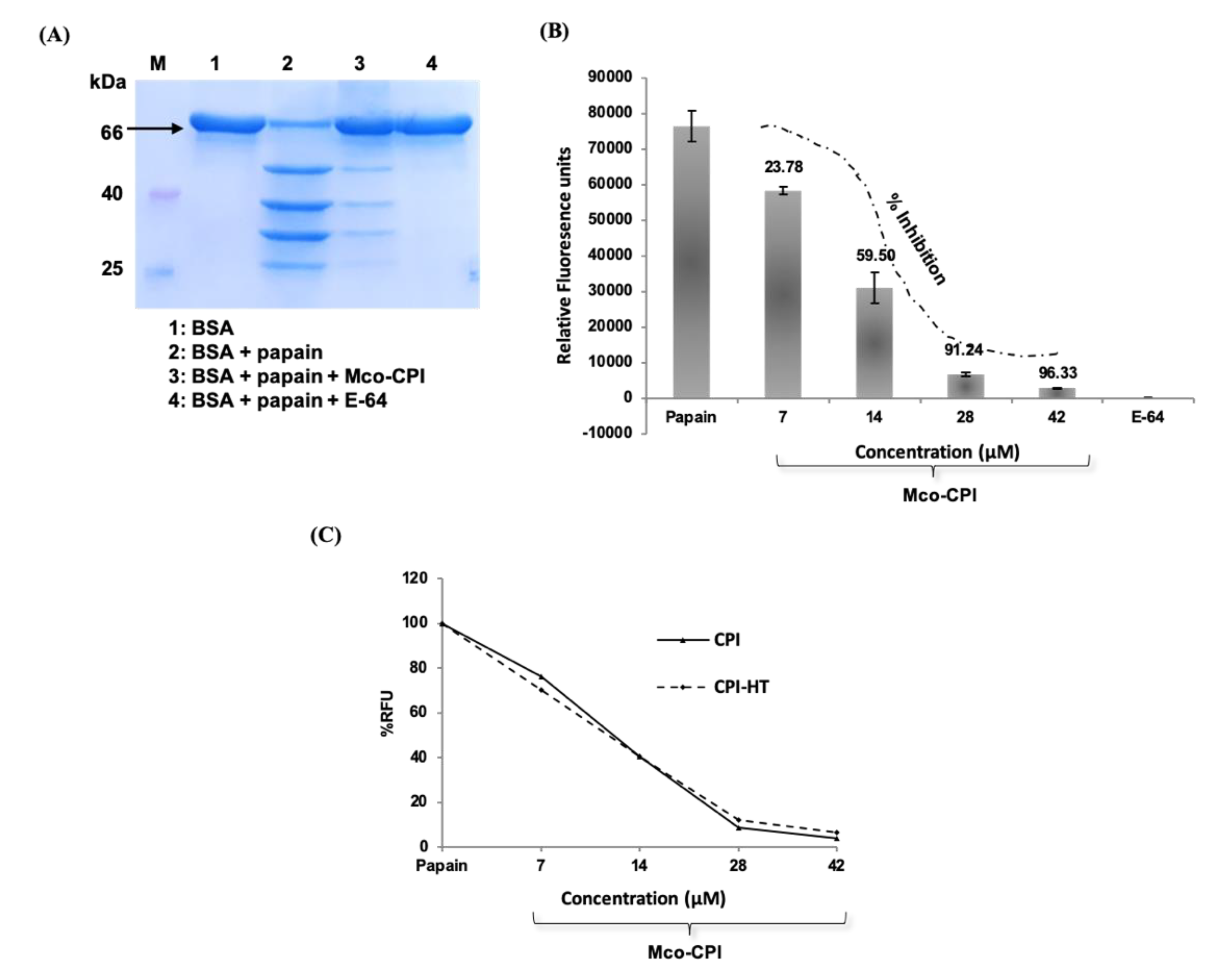
Right-hand side: opportunistic discovery of degrader molecules, whereby a known molecule is shown to have a degrader effect, making it possible to identify the E3 ligase mediating that degradation and determine whether that degrader mechanism could be expanded to target additional proteins of interest. Left-hand side: discovery of PROTACs composed of a target-binding moiety (green), a linker (orange) and a ligase-binding moiety (blue), enabling the rational discovery of heterobifunctional molecules to degrade a desired target. b | Schematic representation of the two foundational modalities for targeted protein degradation. ARV-110 and ARV-471 target the androgen receptor and the oestrogen receptor, respectively, while the E3-binding ligand interacts with the cereblon (CRBN) E3 ligase. PROTACs are composed of a target-binding moiety (green), a linker (orange) and an E3 ligase-binding moiety (blue). It is this catalytic-type mechanism of action (MoA) and event-driven pharmacology that distinguishes PROTACs from classical inhibitors, which have a one-to-one relationship with the POI and whose pharmacology is driven by stoichiometry and, usually, by interactions with a catalytic site.Ī | Structure and properties of two proteolysis-targeting chimeras (PROTACs) that have entered clinical trials, ARV-110 and ARV-471. Simultaneous binding of the POI and ligase by the PROTAC induces ubiquitylation of the POI and its subsequent degradation by the ubiquitin–proteasome system (UPS), after which the PROTAC is recycled to target another copy of the POI (Fig. These are heterobifunctional small molecules consisting of two ligands joined by a linker: one ligand recruits and binds a protein of interest (POI) while the other recruits and binds an E3 ubiquitin ligase. Many of these targets have key roles in cancer and other diseases, and so have remained of great therapeutic interest, despite their recalcitrance to small-molecule inhibitors.Ī major class of molecules that may enable such proteins to be modulated through TPD are known as proteolysis-targeting chimera (PROTAC) protein degraders. Some have been intractable because their active sites are broad, shallow pockets that are difficult to bridge with small molecules others have ‘smooth’ surfaces that offer few sites for a small molecule to bind. Targeted protein degradation (TPD) is attracting substantial interest owing to its potential to therapeutically modulate proteins that have proved difficult to target with conventional small molecules. We then discuss key areas for the future of TPD, including establishing the target classes for which TPD is most suitable, expanding the use of ubiquitin ligases to enable precision medicine and extending the modality beyond oncology. In this Review, we summarize the first two decades of PROTAC discovery and assess the current landscape, with a focus on industry activity. With clinical proof-of-concept for PROTAC molecules against two well-established cancer targets provided in 2020, the field is poised to pursue targets that were previously considered ‘undruggable’. In the 20 years since the concept of a proteolysis-targeting chimera (PROTAC) molecule harnessing the ubiquitin–proteasome system to degrade a target protein was reported, TPD has moved from academia to industry, where numerous companies have disclosed programmes in preclinical and early clinical development. Targeted protein degradation (TPD) is an emerging therapeutic modality with the potential to tackle disease-causing proteins that have historically been highly challenging to target with conventional small molecules. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
